We invite any Government, any scientist and HIV groups for collaboration to reach out this invention to 33 million HIV people globally
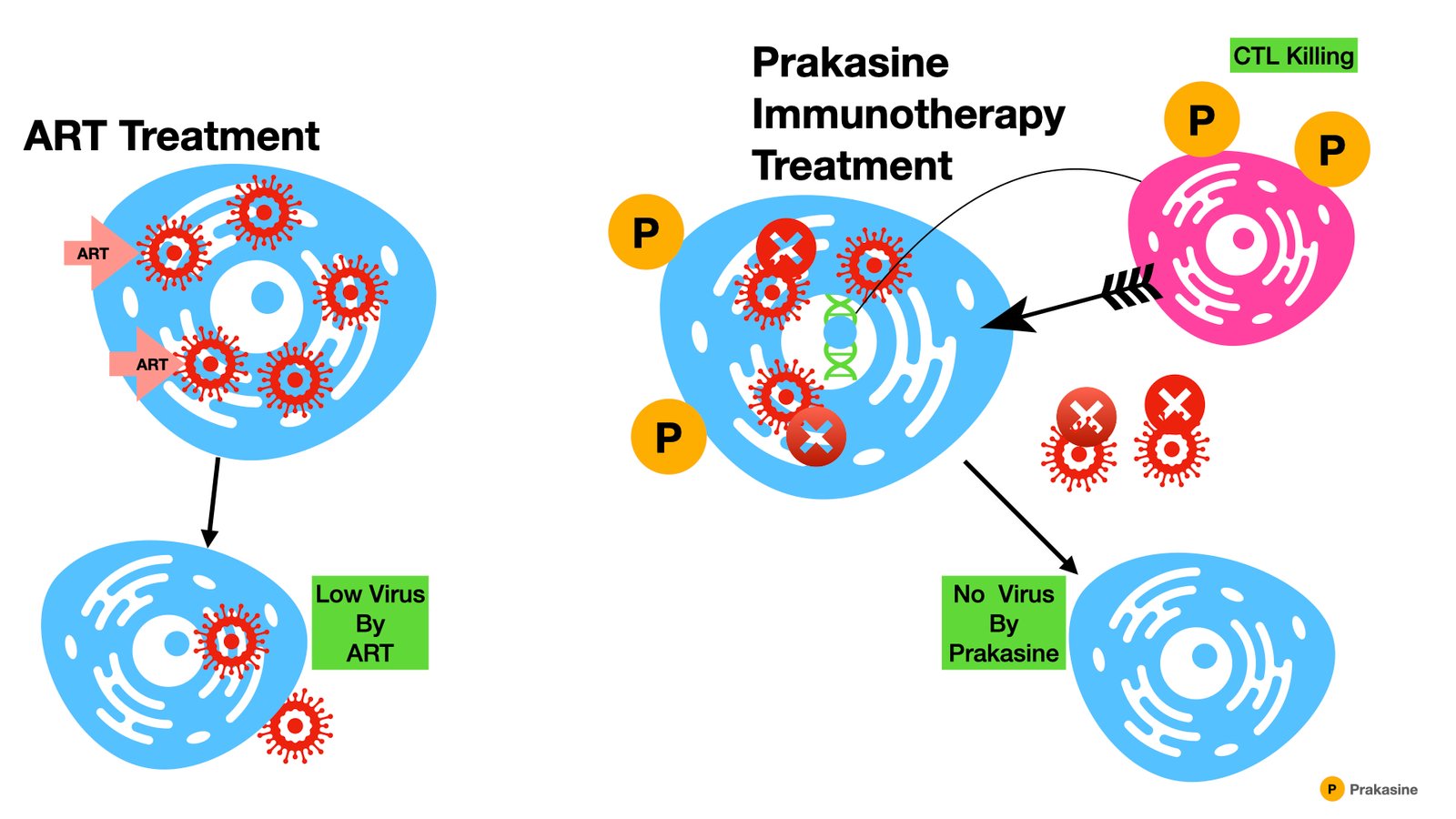
Prakasine is the immunotherapy developed by the Indian Scientist Dr. SK Prakash by developing the organometallic nanoparticle to stimulate the immunostimulatory, immunomodulatory, and immunoregulatory cytokines gene expressions to increase the natural immunity, adaptive immunity, and cell-mediated immunity together simultaneously. So this Prakasine can increase the immunity in HIV, Cancer, and other infectious disease patients.
Latest HIV Cure News in the world now and Today
As of now, HIV-affected individuals need lifelong treatment in the entire world scenario except for our Naval AIDS Research center HIV patients because our patients are enjoying our treatment without any side effects. In general, the Highly active antiretroviral therapy (HAART) is being administered to HIV patients all over the world. Even though the highly active antiretroviral therapy improves the quality of life of people living with HIV, the treatment has to be continued lifelong. As there is no permanent cure or some remission from the treatment/drugs the HIV patients are suffering from drug toxicities, interaction with other drugs and persistence of inflammation, and acceleration of the aging process in addition to cost of treatment. Also, HAART produces daunting effects like loss of functions of vital organs such as kidneys, liver, heart, bone marrow, and nervous system.
The advent of highly active antiretroviral therapy came in 1996 but I started my research in 1993 with our Tamil Traditional medicine with the ailing HIV patients in and around Namakkal locality and I could save some patients even though I could not cure them completely. Before that, I have been watching all the patients dying of treatment failures. In India, the ART came in the year 2003. The ART made breakthroughs in the management of HIV disease to the extent of low level or undetectable viremia. However, at any time, these individuals would be likely to go for a viremia when these individuals either stop the treatment or attain resistance to antiretroviral therapy (ART).
The main barrier to a cure is the presence of a “latent reservoir;” a population of HIV-infected cells will persist for the lifetime of the individual despite ART- and the HIV-specific immune response. Eliminating the viral reservoir or making the reservoir inactive forever would prevent the virus from reestablishing the infection after the discontinuation of the treatment. Hence, the identification of various sources of viral reservoirs such as latent CD4 cells, central nervous system – microglial cells, macrophages, monocytes – (peripheral blood mononuclear cells), reproductive system, lymph nodes, spleen, and gut tissues get the prime importance in the research (1).
There are previous HIV Cure reports made by the other scientists and doctors all over the world by employing various approaches like Allogeneic hematopoietic stem cell transplantation from a donor homozygous for HIV-resistant CCR5Δ32 mutation (2), alteration of the host genome using genetic engineering techniques, Kick and kill (also known as “shock and kill”), altering the immune system for better clearance of the HIV-infected targets, “Hit early and Hit hard” (early treatment) are some of the strategies (3).
Potential HIV Cure with Prakasine immunotherapy
Our Prakasine immunotherapy alone can do better than all the other HIV cure strategies (4). Also, it can make tremendous changes in the HIV patients’ life if it is combined with all the above strategies.
1. Sankaranantham M. HIV – Is a cure possible?. Indian J Sex Transm Dis AIDS. 2019;40(1):1-5. doi:10.4103/ijstd.IJSTD_112_15
2. Hütter G, Nowak D, Mossner M, Ganepola S, Müssig A, Allers K, et al. Long-term control of HIV by CCR5 delta32/Delta32 stem-cell transplantation. N Engl J Med. 2009;360:692–8. [PubMed] [Google Scholar]
3. Pace M, Frater J. A cure for HIV: Is it in sight? Expert Rev Anti Infect Ther. 2014;12:783–91. [PubMed] [Google Scholar]
4. Prakash SK (2020) Immunological and Virological Effects of Novel Prakasine Nanomedicine in HIV-Infected Patients in South India: A Preliminary Study. J Virol Antivir Res 9:2. doi: 10.37532/jva.2020.9(2).195
